Biorad western blot protein ladder4/10/2024  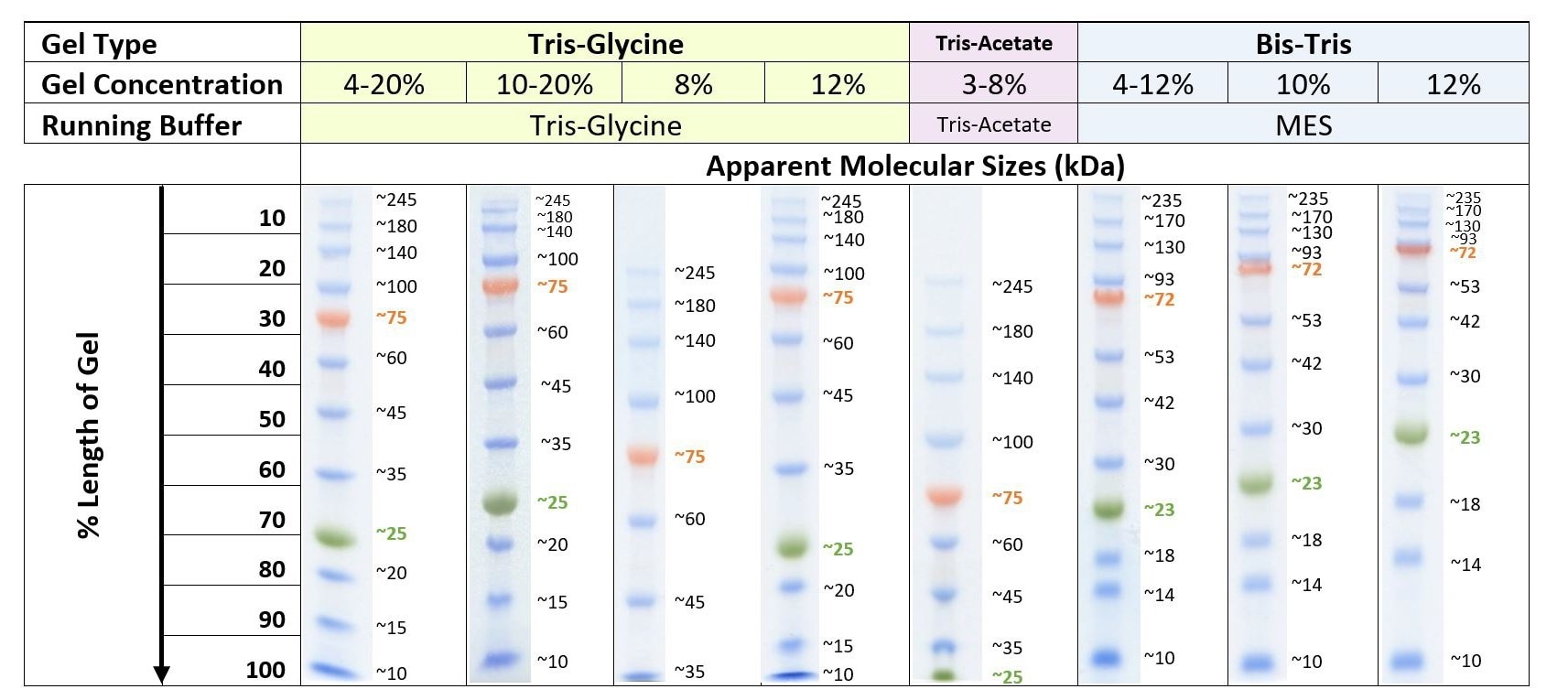
A similar charge-to-mass ratio for all proteins in a mixture, since SDS binds at a consistent rate of 1.4 gram of SDS per gram of protein.Since SDS is negatively charged, it masks the intrinsic charge of the protein and provides an overall negative charge to the proteins.In addition, SDS binds noncovalently to proteins and imparts several characteristics that can be exploited for protein separation: When proteins are separated in the presence of SDS and denaturing agents, for example, reducing agents to dissociate intra-protein disulfide bonds, they become fully denatured and dissociate from each other. This method incorporates the detergent sodium dodecyl sulfate (SDS) into the buffer system and has become the most popular form of protein electrophoresis. Individual proteins are separated into bands according to their molecular weights. Moreover, because the native charge is preserved, proteins can migrate towards either electrode depending on their charge. Additionally, since protein-protein interactions are retained during separation, some proteins may also migrate together as multisubunit complexes and move in unpredictable ways. Since the native charge-to-mass ratio is preserved, protein mobility is determined by a complex combination of factors. Native PAGE is a method in which proteins are prepared in nonreducing, nondenaturing sample buffer, and electrophoresis is also performed in the absence of denaturing and reducing agents. Schematic of electrophoretic protein separation in a polyacrylamide gel. If the gel is run for too long, the proteins can run off the gel. The movement of the proteins through the gel is normally tracked visually by the addition of a dye to the sample, which can be seen moving further down the gel over time. Applying an electrical field across the buffer chambers forces the migration of proteins into and through the gel. Usually, the gel has a vertical orientation, and the gel is cast with a comb that creates wells in which the samples are deposited. In most PAGE applications, the gel is mounted between two buffer chambers, and the only electrical path between the two buffers is through the gel. 
This difference in migration rate between smaller and larger proteins leads to their physical separation in the gel. As proteins move through the gel in response to an electric field, the gel’s pore structure allows smaller proteins to travel more rapidly than larger proteins. This polyacrylamide gel is what lends the name to the technique: polyacrylamide gel electrophoresis, or PAGE. Polyacrylamide Gel Electrophoresis (PAGE) OverviewĪlmost all methods for protein separation use an aqueous polyacrylamide gel as a size-selective sieve during separation. Addition of detergents or reducing agents.Protein size, shape, and charge - which are in turn affected by.Gel composition and strength (weight percentage of polymer).The rate proteins move in gel electrophoresis is governed by relationships among the characteristics of the electrophoresis system and the proteins.įactors affecting protein migration include: In protein electrophoresis, the proteins move through and aqueous polymer gel, which retards migration and retains them for imaging and analysis. These factors allow for differences in protein migration rates to be tuned to a degree, thus enhancing separation. Protein conformation can also be changed by buffering, detergents, and oxidation/reduction. Because the mass of a protein is constant, its charge-to-mass ratio is thereby altered. 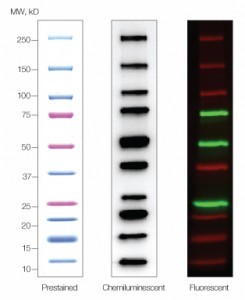
The total charge on a protein in an aqueous medium depends on the degree of ionization of its acid and base groups, which can be controlled by buffering. As a result, proteins have characteristic migration rates that can be exploited for separation. Proteins have a wide range of sizes and shapes and have charges imparted to them by their constituent amino acids. In an electric field, proteins move toward the electrode of opposite charge. Electrophoresis has been developed into a standard molecular biology laboratory technique, where molecules of different sizes and charges are transported through a medium at different rates by an applied electrical field, resulting in their separation. The term electrophoresis refers to the movement of charged molecules in response to an electric field. Electrophoresis Basics How Protein Electrophoresis Works 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
